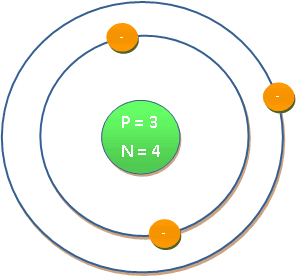
| Atomic number | : | 3 |
| Atomic mass | : | 6.941 g.mol-1 |
| Electronegativity according to Pauling | : | 1.0 |
| Density | : | 0.53 g.cm-3 at 20 °C |
| Melting point | : | 180.5 °C |
| Boiling point | : | 1342 °C |
| Vanderwaals radius | : | 0.145 nm |
| Ionic radius | : | 0.06 nm |
| Isotopes | : | 2 |
| Electronic shell | : | 1s2 2s1 or [He] 2s1 |
| Energy of first ionisation | : | 520.1 kJ.mol-1 |
| Standard potential | : | - 3.02 V |
| Discovered by | : | Johann Arfvedson in 1817 |
Periodic Table of Elements