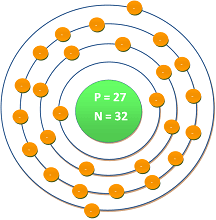
| Atomic number | : | 27 |
| Atomic mass | : | 58.9332 g.mol-1 |
| Electronegativity according to Pauling | : | 1.8 |
| Density | : | 8.9 g.cm-3 at 20 °C |
| Melting point | : | 1495 °C |
| Boiling point | : | 2927 °C |
| Vanderwaals radius | : | 0.125 nm |
| Ionic radius | : | 0.078 (+2) nm ; 0.063 (+3) nm |
| Isotopes | : | 8 |
| Electronic shell | : | [Ar] 3d7 4s2 |
| Energy of first ionisation | : | 757 kJ.mol-1 |
| Energy of second ionisation | : | 1666.3 kJ.mol-1 |
| Energy of third ionisation | : | 3226 kJ.mol-1 |
| Standard Potential | : | - 0.28 V (Co2+ / Co) ; + 1.84 V (Co3+ / Co2+) |
| Discovered by | : | George Brandt in 1737 |
Periodic Table of Elements